Administering medications via intravenous (IV) route involves delivering drugs directly into the bloodstream using different methods such as IV push, drug incorporation into IV fluids, or through a volumetric chamber (Soluset). IV push allows medication to be injected directly into the IV line over a short period of time. Drug incorporation involves adding medication to an IV solution for continuous infusion, while a volumetric chamber (Soluset) controls the amount of fluid and medication administered, commonly used in pediatric patients. These methods ensure accurate dosing and rapid therapeutic effect.
Why is this procedure performed?
- To administer medications directly into the bloodstream for rapid action.
- To ensure accurate and controlled delivery of prescribed drugs.
- To provide continuous medication infusion when required.
- To allow administration of medications in patients who cannot take oral drugs.
- To regulate the amount of medication and fluid given, especially in pediatric patients using a volumetric chamber (Soluset).
- To achieve immediate therapeutic effects in emergency or critical situations.
Materials Needed
- 3cc or 5cc syringe
- 5cc syringe
- 10cc syringe
- Sterile water vial
- Cotton ball with alcohol
- Volumetric chamber/ Soluset
- Needle gauge 21/ gauge 18
- Ordered medications
Assessment
- Validate the orders
- Examine the medication administration record for accuracy and completeness
- Review information on the drug including:
- Effects
- Dilution
- Rate of administration
- Potential for incompatibility with other fluids or medications to be given
- Assess for what type of IV access is present
Planning
- Determine the equipment you will need
- Wash your hands
- Select materials needed
- Check placement, patency and backflow of IV line.
Implementation
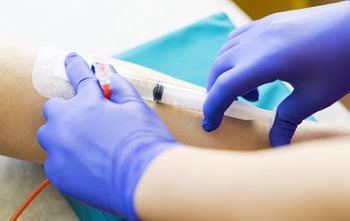
IV Push
- Explain procedure (Name of medicine and action) before administration
- Check the IV site placement, patency & backflow. Check for ANST of the drug for IV push (if applicable)
- Disinfect Y-injection site.
- Kink tubing, pierce through the Y-injection site, and push prepared drug slowly as ordered
- Flush IV tubing after drug administration
- Regulate rate of IVF infusion as ordered
.jpeg)
Incorporation of drug into IVF
- Follow procedure A and B on IV push
- Locate and disinfect the injection port with an alcohol swab. (Medication injection port hangs separately below an IV plastic bag or can be found as the smaller circle on top of rubber cap for an IV plastic bottle.)
- Incorporate prepared drug aseptically.
- Return the cover of injection port, if there’s any.
- Shake the bottle to mix the incorporated medicine with the IV solution.
- Regulate flow rate
- Place an IV label
.jpeg)
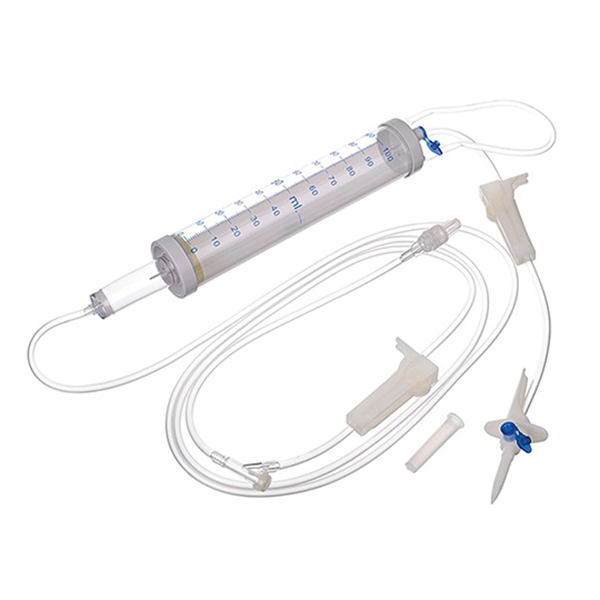
Incorporation into volumetric chamber (soluset)
- Follow procedure A and B on IV push
- Open packaging of the volumetric chamber and IV diluent. Close roller clamps, and spike IV diluent. Hang the IV diluent bottle on the IV pole.
- Fill the volumetric chamber with the ordered amount (ml) of IV diluent by opening the clamp between the IV fluid and the volumetric chamber and adding more diluent for priming the tubing.
- Clamp tubing from the IV bottle and open the clamp of the airway at the volumetric chamber.
- Ensure that the floater is above the IV diluent inside the volumetric chamber.
- Attach a large bore needle at the end of the soluset to be connected to the Y-injection port of the main line.
- Tape the needle cover onto the soluset line.
- Open the clamp positioned after the volumetric chamber. Prime the tubing. The needle should be positioned under a kidney basin.
- Disinfect injection port of the volumetric chamber
- Incorporate prepared drug to the desired IVF as dilution. Mix/swirl the soluset.
- Disinfect the Y-injection port of the main IVF line and insert the needle of the soluset line. Tape both lines, do not cover the injection port.
- Regulate flow rate.
- Place IV label on volumetric chamber
Evaluation
Evaluate using the following criteria:
- Patients’ rights followed
- Correct route used
- Effectiveness of medication assessed
- Any side effect promptly identified
Documentation
Record pertinent data in terms of:
- Medication dosage
- Route of administration
- Time of administration
- Signature
